Herbivory may be changed by climate change and how does that affect the host plants? Find out in the Early View paper “Colonization of a host tree by herbivorous insects under a changing climate” by Kaisa Heimonen and co-workers. Below is their summary of the paper: Climate warming is predicted to increase the abundance of herbivorous insects due to increased survival, growth and multivoltinism. In addition, due to warming climate many insect species are predicted to shift their ranges to higher latitudes. Host plants are adapted to the present day herbivore pressure and insect communities but in the future the abundance of insects and the composition of herbivorous insect communities might change which can lead to more intense herbivore damage. We wanted to study the susceptibility of silver birch (Betula pendula Roth) populations from different latitudes to the insect herbivores that are expected to spread northwards in the future. To do this we established three common gardens with 26 genotypes of silver birch from six latitudinal populations in Finland ranging from 60°N to 67°N. The common gardens were located at three different latitudes 60°N, 62°N and 67°N. At each study site 260 silver birches were growing. This experimental setup is being used also for several other studies (see the project homepage: http://www.uef.fi/fi/birchadaption).
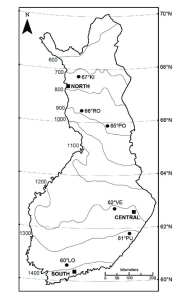
Figure 1. Map showing the three common garden sites (filled squares) and the six source populations (filled circles). Mean annual temperature isoclines are shown in grey.

Figure 2. The three common garden sites in Finland where the study was conducted. A) Southern study site is located in Tuusula 60°N, B) Central study site is located in Joensuu 62°N and C) Northern study site is located in Kolari 67°N. Photo credits: Kaisa Heimonen.
We wanted to study how the local insects at each of the common garden sites colonized the translocated birch genotypes. We asked if the insect herbivore density, species richness or community composition could be explained by the source population of the birch or by the direction or distance of the latitudinal translocation. The herbivore community on the study birches was examined during two growing seasons in 2011 and in 2012.

Figure 3. Kaisa Heimonen (lead author) observing the herbivorous insects on silver birch at the northern study site in 2012. Photo credits: Sari Kontunen-Soppela.
Herbivore density among the source populations differed in 2012 but not in 2011 and species richness was not affected by the source population. Latitudinal translocation could not explain the variation in the herbivore density or in the species richness. Community composition of the herbivores differed among the source populations at two of the three study sites and the similarity of the herbivore communities decreased with increasing latitudinal distance of the source populations.

Figure 4. Common insect species on silver birch belonging to the orders Lepidoptera, Coleoptera and Hymenoptera. A) White-shouldered smudge (Ypsolopha parenthesella), B) Birch leaf roller (Deporaus betulae) and C) Early birch leaf edgeminer (Fenusella nana). Photo credits: Kaisa Heimonen.
Silver birch genotypes from source populations originating from closer geographical distance had more similar herbivore community composition at our experimental sites possibly because they are genetically more similar than the geographically more distant birch genotypes. All birch genotypes were colonized by some of the local herbivores at all three study sites suggesting that in the future herbivorous insects are able to colonize novel host plant genotypes. The results of this study show that compositional changes in the insect communities on their host plants are expected in the future. Newly structured herbivore communities might affect the herbivore damage and thereby also the plant growth.







